
Robert Benezra
Cells with unstable genomes are one of the hallmarks of cancer. While most normal human cells have an identical set of 46 chromosomes, certain gene changes can make cells divide unevenly, leading to the birth of new cells with extra or missing chromosomes, and sometimes chromosomes with altered structure. This phenomenon, known as chromosome instability, is believed to stimulate the formation and growth of tumors.
Now a recent study published in Nature and lead by Robert Benezra, of the Sloan Kettering Institute’s Cancer Biology and Genetics Program, suggests that chromosome instability has additional consequences: It may explain why some patients who initially respond to a targeted therapy — a drug designed to interfere with a specific gene or protein that causes tumor growth or metastasis — eventually relapse, often with new tumors that no longer respond to the therapy.
The investigators used a mouse model of lung cancer, in which blocking the activity of a mutated gene called Kras was sufficient to kill tumor cells. In some of the mice, chromosome instability had been induced artificially in tumors. Experiments showed that a tumor eliminated by blocking Kras was more likely to recur if chromosome instability had been induced.
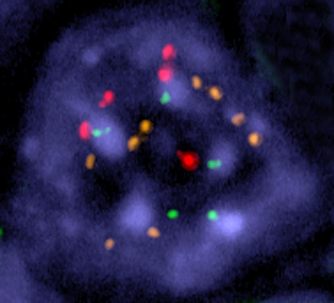
A tumor cell in which chromosome instability has been induced. Multiple copies of three of the cell’s chromosomes (shown as red, yellow, and green dots) are present in the cell nucleus (shown in blue). In a normal cell, each chromosome would be present in two copies. Image courtesy of Rocio Sotillo and Robert Benezra.
Dr. Benezra said the findings have implications for the use of targeted therapies aimed at killing the tumor cells that remain in a patient after a tumor has been removed. [PubMed Abstract] “If the cells’ chromosomes are unstable, such cells might over time be able to resist the therapy we use to eradicate them,” he said. He and his colleagues believe chromosome instability can enhance a cancer’s capacity to evolve and metastasize by generating cells with high genetic diversity.
“Even with our best targeted therapies, which might kill the vast majority of a patient’s tumor cells, an occasional cell might be able to adapt to the type of stress a drug imposes,” Dr. Benezra explained. That cell could then multiply and seed new cancer growth, possibly leading to tumors that are resistant to the therapy.
Dr. Benezra and his colleagues are now exploring the molecular mechanisms that lead to chromosome instability, hoping that the genes or proteins involved could be targeted therapeutically. “Many patients who develop metastatic disease do so after undergoing seemingly successful treatment and being symptom-free for many years,” he added. “The development of therapies that specifically kill cells with unstable chromosomes after initial cancer treatment could one day make it possible to prevent such recurrences.”
