Because treatment can often damage the immune system, people with cancer are remarkably susceptible to infections. This is especially true for those who undergo bone marrow transplants for blood cancers or intensive chemotherapy for certain types of leukemia. Both of these treatments wipe out infection-fighting white blood cells while combating the disease.
Memorial Sloan Kettering researchers have long been at the forefront of studying how the body responds to infections. In addition to the potential benefits for people with cancer, their work has implications for a much broader group. Two studies recently published by MSK scientists are showing that the best way to fight dangerous bacteria may be with other, nonharmful bacteria.
“These protective species of bacteria were not completely unknown to us, but they were not well characterized,” says physician-scientist Eric Pamer, an infectious diseases specialist, Head of the Division of Subspecialty Medicine at MSK, and the senior author of both studies. “They’re what we call commensal bacteria. They reside within us but do not cause disease. They’re primarily little workers that help us process and digest our food. And it turns out that some of them can also protect us from infections in the gut.”
Antibiotics are an important part of treatment for many people with cancer. They can, however, sometimes wipe out populations of these beneficial bugs.
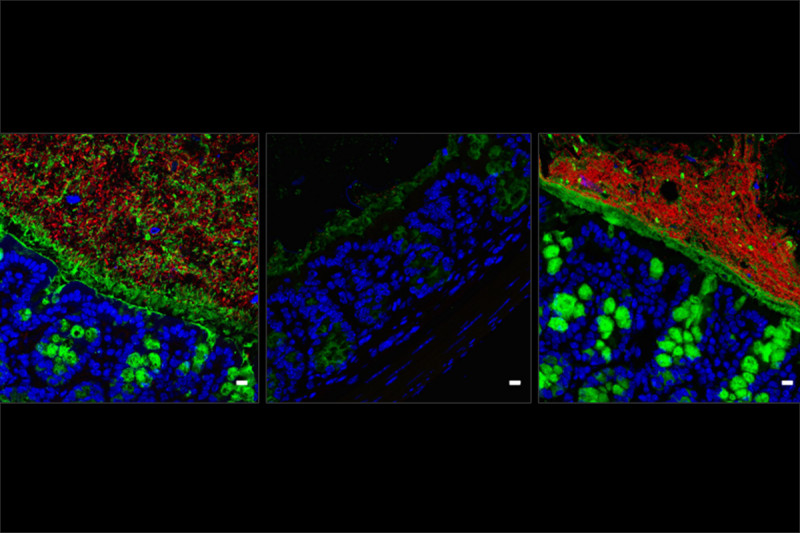
A study published in Cell Host Microbe discovered strains of bacteria that protect against vancomycin-resistant Enterococcus (VRE). VRE is a potentially deadly infection that is frequently observed in hospitalized people. The other paper, published in the Journal of Experimental Medicine, investigated species that protect against Listeria monocytogenes. Listeria is a food-borne bacteria that is particularly dangerous for people with cancer, the elderly, and pregnant women. When these infectious microbes colonize the intestines, it can lead to serious complications.
Microbe Hunters
“Our latest finding with VRE builds on research that we’ve been doing for more than a decade,” Dr. Pamer explains. “We’ve long suspected that particular bacterial species protected people from VRE colonization in their gut, but until now we didn’t know which species.”
In the Cell Host & Microbe study, former graduate student Silvia Caballero, the paper’s first author, studied mice that were able to fight off VRE. She compared them with mice that were highly susceptible to VRE infections. She employed DNA sequencing to compare the microbiomes of the two types of mice. (A microbiome includes the genes of all the microorganisms that naturally inhabit the body.) By using this approach, Dr. Caballero was able to identify some particular species of bacteria present in the mice that resisted infection and absent in mice that were vulnerable to it.
In the other study, postdoctoral fellow Simone Becattini used a similar tactic to identify certain bacterial species that help mice resist infection with Listeria.
This type of microbiome sequencing and analysis is not entirely new to Dr. Pamer’s lab. In 2012, his team, in conjunction with investigators in MSK’s Computational and Systems Biology Program, conducted another study that identified which species of bacteria offered protection against Clostridium difficile, another infection frequently acquired by hospitalized patients.
Restoring Good Bacteria
People with cancer often lack these protective bacterial species because the antibiotics that are an important part of their treatment kill the strains off. Broad-spectrum antibiotics, which many patients receive, fight a wide range of bacteria but can also damage helpful kinds. Some experts have pointed to more careful use of antibiotics to prevent these types of infections.
Dr. Pamer suggests that other methods are more likely to be successful. “As an infectious disease practitioner treating people at a cancer hospital, I see a lot of patients who are profoundly immunocompromised because they have no white blood cells in circulation,” he explains. Essentially, the antibiotics are keeping them alive. “A preferable route, and one that we are beginning to investigate here, is to treat people with whatever antibiotics are needed while at the same time recognizing that we are damaging their commensal bacteria and will need to replenish them later.”
Several approaches for restoring commensal bacteria are being explored at MSK. One proposal is to create probiotic treatments using these newly discovered protective species. The missing bacteria would then be given back to patients. However, unlike the probiotics that are present in yogurt and other foods, or those sold as supplements at health food stores, the bacteria that protect against VRE, Listeria, and C. difficile can’t survive in air. They would need to be administered in a different way, such as in a sealed capsule.
Researchers are also looking at the products these protective bacteria secrete. Some of these products confer protection and could potentially be developed to treat people with infections.
MSK physician-scientist Ying Taur in the Infectious Diseases Service and physicians from the Bone Marrow Transplant Service are investigating another approach called an autologous fecal transplant. Patients’ microbiota are collected and stored before they undergo bone marrow transplants. After cancer treatment is complete, the microbiota are readministered to the patients.
“At MSK, we’re really in a remarkable position to do this kind of research,” Dr. Pamer concludes. “Our infectious diseases group has been able to exploit the clinical data we’ve collected on all these patients over the years. Our experts in pharmacy, chemistry, hematology, and microbiology — as well as our computational biologists — are helping us understand what’s driving the expansion of these antibiotic-resistant bugs. All of this research is likely to make a real difference for patients here and around the world.”