There are many effective prostate cancer drugs, but a major challenge is that they often stop working. One common treatment, hormone therapy, is initially effective at blocking the cancer-fueling effects of testosterone. But these drugs can lose their power as the prostate cancer evolves.
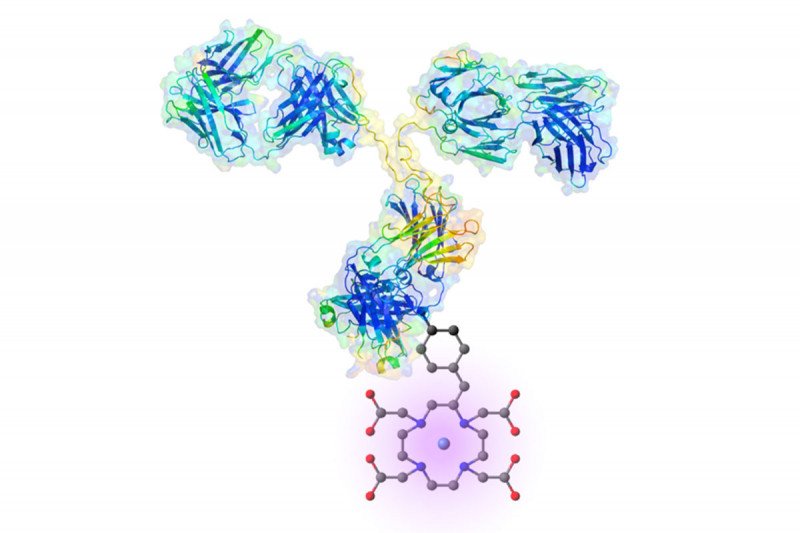
Researchers have looked for better ways to target prostate cancer cells and monitor whether treatments are working or the cancer is progressing. In 2016, promising news emerged from the laboratory of Steven Larson in the Sloan Kettering Institute of Memorial Sloan Kettering. Scientists demonstrated that an antibody called hu11B6 binds to a protein, called hK2, which is secreted only by prostate cells.
In that study, the researchers tested this technology for diagnostic purposes, proving in mice that they could track prostate cancer spread and determine when hormone therapy was no longer working. Now these same researchers report that this approach can be used to deliver drugs to prostate cancer cells without destroying nearby healthy cells.
By linking hu11B6 to a radioactive particle called actinium-225, they showed they could deliver a precision strike to the prostate cancer cells. In mice, a single dose of the drug wiped out prostate cancer and caused the animals to live significantly longer. The researchers further showed that hu11B6 is efficient in nonhuman primates. The distribution of hu11B6 — how it moves from the bloodstream into the body’s tissues — is the same whether carrying a diagnostic particle or a toxic warhead such as actinium-225. This makes it possible to plan the minimal dose needed to treat a patient.
The method is reported today in the journal Nature Communications by molecular pharmacologist Hans David Ulmert and radiochemist Michael McDevitt, along with other researchers at MSK and several other institutions.
“Unlike radiation or chemotherapy, this therapy offers the possibility of exclusively targeting prostate cancer cells and represents a potential improvement in diagnosis, staging, and treatment,” Dr. Ulmert says. “Because we can choose which radionuclide to attach to hu11B6, we have exquisite control over the lethality of the drug and its risk to patients.”
Enlarging the Target
One advantage of this therapeutic approach is that, in effect, it makes the target more abundant as it works. Production of hK2 is regulated by a protein called an androgen receptor, which plays a central role in driving prostate cancer. When the radiolabeled antibody is drawn into the cell by binding to hK2, the cells are either immediately wiped out, or repair mechanisms in the cells set off a cascade of events that makes the androgen receptor more active. This in turn boosts hK2 production, giving the drug even more hK2 to home in on.
The research team was recently awarded the Impact Award by the Department of Defense to develop and evaluate non-invasive imaging technologies to assess this radiobiological phenomenon.
Dr. Ulmert is optimistic that this technology could enter human testing soon. A humanized form of the hu11B6 antibody has already proved to be safe in nonhuman primates. A clinical trial is currently being planned that will be led by medical oncologist Michael Morris and conducted at MSK.
“This could launch a wave of new antibody-based drugs targeting the hK2 protein,” Dr. Ulmert says. “Given that a single injection could eradicate the cancer, it strongly suggests we are hitting the right target. If we are successful at using this approach to deliver drugs in humans, it could change the course of the disease.”


