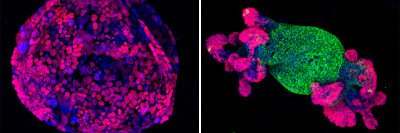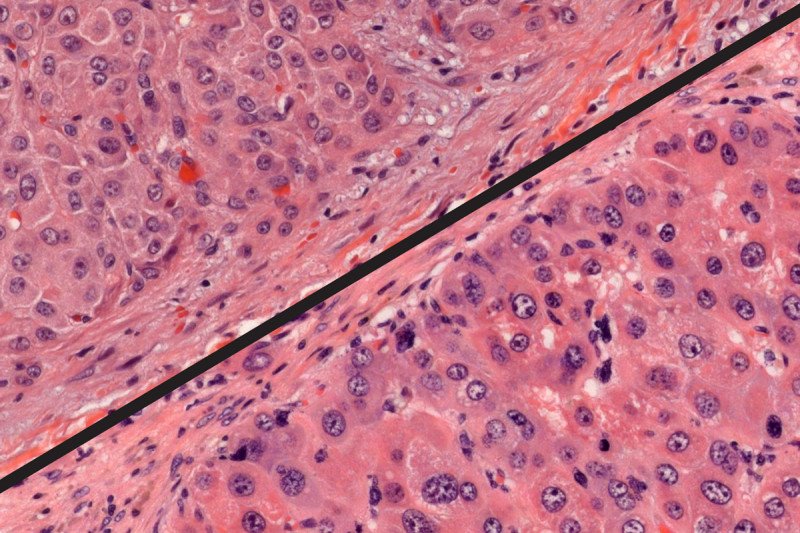
Under a microscope, a genetically engineered mouse tumor (bottom) appears remarkably like a human FL-HCC tumor (top).
Some diseases are rare. Others are ultrarare. But if you have an ultrarare condition, that qualifier is little consolation.
That’s certainly the case for people with an uncommon type of liver cancer called fibrolamellar hepatocellular carcinoma (FL-HCC). It strikes fewer than 1,000 individuals a year worldwide, mostly children and young adults. There are few effective treatments besides surgery. FL-HCC is usually diagnosed late, so it is often fatal. The five-year survival rate is just 34%.
Because FL-HCC is so rare, it’s hard for researchers to learn more about it, or even to conduct clinical trials of potential medicines. But a new advance, pioneered by scientists at MSK, could provide a powerful way to decipher the disease’s underlying biology.
Using the genome-engineering tool CRISPR, a team of researchers led by molecular biologist Scott Lowe developed a mouse model of FL-HCC that reliably reproduces many aspects of the human cancer. The model should make it easier for scientists to develop and test drugs that could be used to treat the disease.
Using CRISPR to Prove a Mutation’s Role
The research came about through a collaboration between Dr. Lowe’s lab at MSK and Sanford Simon’s lab at The Rockefeller University. Several years ago, Dr. Simon and his team discovered that all people with FL-HCC invariably share the same genetic alteration: a mix-up in liver cells that causes two genes to fuse together. This observation suggested that this mutation was somehow responsible for the cancer. It was a big advance — one made even more remarkable by the fact that Dr. Simon’s own daughter, Elana, had suffered from FL-HCC and was centrally involved in the research effort.
But still, the result was only a correlation.
To find out for sure whether the mutation was responsible, the teams decided to try to recreate the genetic abnormality in a mouse. Dr. Lowe’s team, including graduate student Edward (Ted) Kastenhuber, used CRISPR to snip out the portion of the chromosome that is lost in people with FL-HCC. The remaining bits of chromosome then combine, fusing two genes that are normally separate.
“Using technologies developed here at MSK, we showed that if you can reproduce that genetic event in mice, they will develop a cancer that looks very much like human FL-HCC,” Dr. Lowe says. “This demonstrates that the mutant gene is causally responsible for FL-HCC.”
A Useful Model for Testing Drugs in FL-HCC
In addition to confirming the role of the mutation, called DNAJB1-PRKACA, in driving the cancer, the team also identified other genetic and environmental factors that could be involved.

Rockefeller University biophysicist Sanford Simon, together with his daughter, Elana, observed that all people with FL-HCC share a common genetic mutation.
Using data from MSK-IMPACT™, a DNA-sequencing test used on tumors, they uncovered another common genetic change found in some people with FL-HCC. When they combined this change with the fusion mutation, the cancer’s growth sped up. This makes the model even more useful, since the researchers don’t have to wait as long for the cancer to develop.
The team plans to test a variety of drugs in the model to see if they can slow or stop the cancer’s growth. FL-HCC could be a good target for drugs called kinase inhibitors, but scientists won’t know for sure until they test them.
“Models give us the freedom to fail and to explore a wide variety of approaches,” Mr. Kastenhuber says. “This way, we can accelerate the discovery of treatments that are more likely to be effective before exposing people to experimental medicines.”
A paper describing the breakthrough appeared in the Proceedings of the National Academy of Sciences. Dr. Lowe is a newly elected member of the academy, and this paper is his inaugural article, meant to highlight special work coming out of an academy member’s laboratory. Dr. Lowe is Chair of the Cancer Biology and Genetics Program at Sloan Kettering Institute and Chair of the Geoffrey Beene Cancer Research Center at MSK.